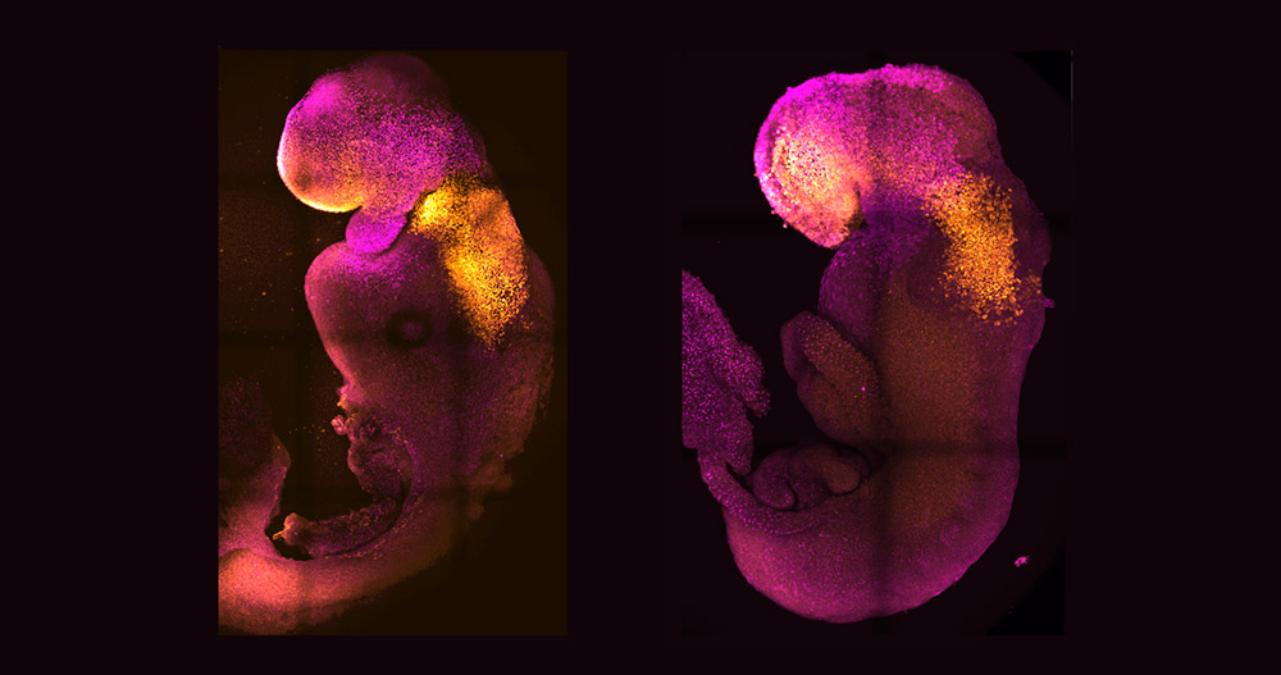
Without an egg cell or uterus, two research teams have grown mouse embryos in a “test tube” for the first time. Instead of fertilized eggs, they used single stem cells. The cultures of stem cells turned into a yolk sac, which was the beginning of the placenta and the embryo. The embryo grew and changed until it had heart cells that beat and the beginnings of all organs, including the brain and intestine. For the first time, the most important steps of embryonic development were completed outside of the womb and without fertilized eggs.
One fertilized cell may produce new life, and this process is very intricate and a wonder of nature. Numerous genetic, pharmacological, and mechanical cues ensure that early embryonic cells travel to the appropriate location and differentiate into the tissue type needed there.
Complex Procedures
However, in order for the embryo to grow and nest, the fertilized egg cell must also create the placenta, which subsequently takes over the supply together with maternal tissue, as well as the yolk sac, which feeds the embryo for the first few days. One embryonic and two extraembryonic cell types are differentiated into at the blastocyst stage, a few days after conception.
This stage is followed by the gastrulation of the embryo with the formation of the cotyledons and, as it progresses, the formation of the first organ precursors, including the neural tube, which gives rise to the brain and spinal cord. If anything goes wrong at this stage, the pregnancy will fail since it lays the scene for everything else that will occur later.

Stem Cells Instead of Fertilized Egg Cells
This highlights how crucial it is for medicine to be able to research this early stage of embryonic development, for instance by simulating this procedure in the lab. Not only observing how the fertilized egg cell develops, but also attempting to recreate the processes using a synthetic embryo is the greatest method to pinpoint the governing mechanisms. This embryo develops from individual stem cells rather than via fertilization.
For the first time, two research teams have now been successful in creating an artificial embryo from stem cells. Three distinct kinds of mouse stem cells were used as the beginning material, and they were combined in specially designed rotating culture vessels. The researchers mimicked the natural forces in the womb and stimulated the cells to continue to grow into the two extraembryonic tissues and an embryo by turning on certain genes in a specially modified culture medium.
Embryo With Placenta and Yolk Sac
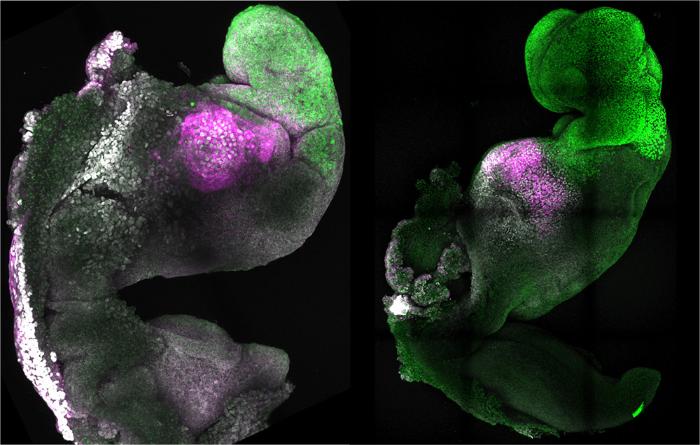
Success was achieved: the originally disorganized stem cells underwent the same phases as real mouse embryos, resulting in the formation of a blastula, a yolk sac, and the placental precursors. The body axis became evident and gastrulation started after around five days. At this point, Zernicka-Goetz’s team observed that the embryos resembled a natural gastrula, and their sizes varied a bit more.
A closer look found that extraembryonic cells act as a key at this early stage, influencing the growth and differentiation of embryonic cells by sending them vital chemical and mechanical signals. This phase of human existence is so mysterious and scientists now examine how stem cells communicate and what may go wrong.
Brain, Heart, and Precursor Organs
More significantly, the synthetic embryo was grown by both study teams for the first time to the stage when organ production starts. According to Zernicka-Goetz, the mouse embryo model not only generated a brain but also a beating heart and all the other components that make up the body. The embryo had attachments for its organs, spine, digestive system, and all of its brain by the eighth day, which was still less than half of its complete maturation period. In the back of the embryo, germ cell precursors were even formed.
The researchers have been working to duplicate these phases of embryonic development for more than ten years, and they have finally been successful. Gene activity and cell metabolism research revealed that the embryos developed from stem cells essentially matched their natural counterparts. Minor variations were seen only in a few kinds of extraembryonic cells.
A second team headed by Jacob Hann of the Weizman Institute of Science in Israel, concluded that despite these variations, the embryos, which are little over eight days old, are very comparable to their natural counterparts, whether they developed within or outside the uterus.
Huge Potential for Science
The findings from these experiments provide crucial insights into early embryonic development and are now creating new opportunities, such as the ability to investigate the origins of malformations. The stem cell embryos are significant because they allow us access to developmental phases that ordinarily take place in the womb. Scientists can now modify individual genes, for instance, to better understand their function in embryonic development, thanks to artificial embryos.
The Moral Issues
But the technique, which has only been used for mouse embryos thus far, also has potentially explosive ethical implications. This is due to the fact that the two research teams are already modifying the processes for using human embryos. This may enable the development of human embryos only from stem cells, devoid of eggs, sperm, or the uterus. While Zernicka-Goetz and her colleagues view this as a chance for, for instance, targeted organ donor breeding, others worry about ethical ramifications:
What is the capacity of a roller bottle for (human) organogenesis compared to a uterus? Many individuals have moral concerns about the prospect of human reproductive cloning, which might result from the use of artificial embryos developed solely from stem cells. This might simplify germ line interventions and the possibility of genetic modifications to the embryo. However, there are a few methodological hurdles that must be overcome before this may occur.
In comparison to a uterus, how far may (human) organogenesis go in a roller bottle? For instance, artificial embryos created only from stem cells might lead to human reproductive cloning, which many people find to be an ethical disaster. This might also make genetic alterations to the embryo and therefore interventions in the germ line easier. Before this to happen, however, a number of methodological obstacles would need to be overcome.
- (Nature, 2022; doi: 10.1038/s41586-022-05246-3; Cell, 2022 doi: 10.1016/j.cell.2022.07.028)