The key points at a glance
- An incandescent light bulb gets its light from a filament that is heated by an electric current.
- The filament is housed inside a glass bulb filled with an inert gas mixture to prevent it from overheating and burning out.
- The incandescent light bulb converts just 5% of the electricity into light, while the other 95% is used to warm the room around it.
An electric current flows through the filament of an incandescent lamp, which is typically a carefully wound tungsten wire, to produce light. Consequently, the filament gives out light (light emission). Its internal composition is shown above. Both the base contact and the foot contact carry electricity to the filament. Filament reaches temperatures during operation range from 4500 to 5400 degrees Fahrenheit (2500 to 3000°C). A specific gas, a noble gas-nitrogen combination, is pumped into the glass bulb around the filament to prevent it from burning out. This is why the light it gives out is orange. A lot more blue light reaches us from the Sun since its surface temperature is roughly 9,900°F (5,500°C).
However, only approximately 5 percent of the electricity fed into an incandescent lamp is turned into light; the other 95 percent is wasted as heat. Depending on its size and construction, an incandescent lamp’s luminous effectiveness may range from roughly 8 lm/W to 20 lm/W. (lm: lumen, unit of luminous flux). General-purpose incandescent light bulbs have a lifespan of roughly 1,000 hours.
Single and double helix filaments

The tungsten filament of an incandescent light bulb is shown coiled and is occasionally coiled twice to produce a double filament. This is due to two factors:
The full length of the about 0.030 mm diameter tungsten wire, which is around 1.0 m long, must fit within the light bulb.
Keeping the wires as close together as possible so that they heat each other is an effective way to keep the heat created in the wire, which is required to reach the high annealing temperature, from being lost too rapidly to the outside as heat loss.
Melting a wire filament
The chemical interaction between the wire and the oxygen in the air that causes the filament to burn through is called oxidation. There is a necessary minimum temperature for oxidation processes to occur. When the wire of metal begins to glow orange, the temperature has risen over the safe threshold. After then, it “burns” due to rapid oxidation by ambient oxygen.
The same thing occurs when oxygen comes into contact with the tungsten filament of an incandescent light bulb.
Preventative gassing to avoid combustion

Pumping the air out of the glass bulb or filling it with a gas that will not react with the filament at these high temperatures is how incandescent light bulbs prevent the filament from oxidizing.
Even if the air is sucked out of the bulb, the hot filament will eventually cause the glass to shatter due to the evaporation of the metal within. Individual atoms may break away from the wire surface at the high temperature of the white-hot filament, which is near the melting point of the wire material. The atoms partly deposit themselves as a black film on the interior of the glass bulb. However, this causes the wire to gradually thin down until it finally snaps. The evaporation process at the glow wire is shown in a picture animation below.
The evaporation of the metal is reduced if a gas of a specific pressure is present surrounding the wire, since this gas prevents the metal atoms from leaving the wire’s surface. These days, inert gases are often used with a little percentage of nitrogen added to increase their pressure (for example, argon with roughly 10% nitrogen).
Lifetime
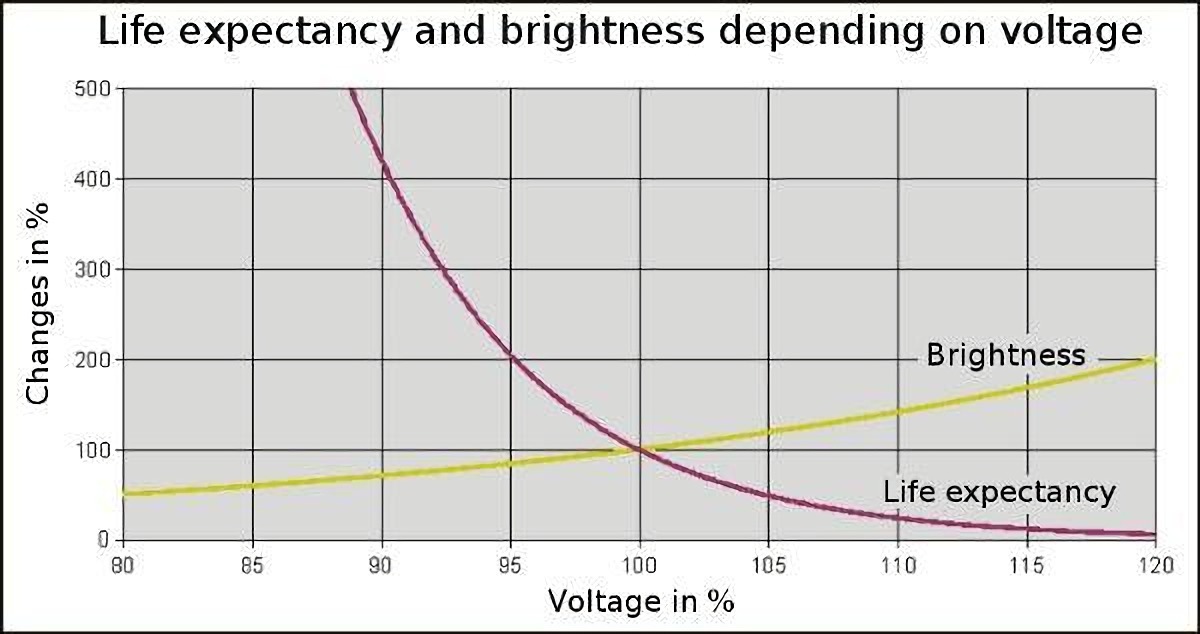
Modern incandescent light bulbs are designed to last for 1000 hours. This is a middle-ground solution since it requires a trade-off between low temperature (lower luminous effectiveness and longer life) and higher temperature (more luminous efficacy and shorter life).
The figure depicts the correlation between an incandescent lamp’s luminous effectiveness (brightness or luminous flux), useful life, and operating voltage. Even a little drop in operating voltage will diminish the lamp’s brightness by a noticeable amount, but it will give you far longer use out of the bulb.
Incandescent light bulb current flow
Upon first activation, an incandescent lamp’s filament can handle a high current since it is cold. However, the wire becomes very hot due to the current flow, which increases the resistance of the filament and slows down the current flow. Therefore, the current is much lower while the device is running compared to when it is first turned on. Because of this, an incandescent light bulb cannot be considered an OHM resistor.
Halogen lamps

An improvement upon the incandescent light, the halogen lamp (above) uses a tungsten filament and a filling gas that includes a halogen component (halogens are iodine, bromine chlorine, etc.). The little glass bulb can be built out of either quartz or tempered glass. Halogen incandescent lights with iodine were introduced in 1958.
Chemically less aggressive and colorless bromine compounds like bromomethane (CH3Br) were developed later, enabling machine manufacture. At lower temperatures near the bulb, the bromine produced from the bromine compound combines with the tungsten atoms evaporating off the filament during operation. Even at temperatures in excess of 480°F (250°C) within the bulb, the tungsten bromide does not settle on the bulb’s wall and instead stays in gaseous form.
This is why the size of the glass bulb is restricted. Close to the filament, the tungsten complex breaks down into atomic tungsten, which binds to the hottest (and hence thinnest) sections of the tungsten wire, and bromine, which recombined to produce bromomethane. Regenerating the filament at its weak areas and preventing bulb blackening from tungsten deposits are also byproducts of this cyclical operation.
As a consequence, greater filament temperatures may be used than in conventional incandescent lights, leading to greater luminous efficacies. Despite their compact size and constant high light output, halogen bulbs have a relatively long lifespan.
Readiness for comprehension

An overhead projector’s controls often include an “economy” and “brightness” switch. Comparing the economy circuit’s 90% operating voltage to that of the typical bright circuit’s 100% operating voltage reveals that the former is much more efficient.
Using, we can find out how much longer the lamp lasts and how much the luminous effectiveness drops as a consequence of this.
The lamp’s useful life is increased by a little over 400% when the operating voltage is decreased by 10%. As a result, the bulbs’ lifespan is increased by more than a factor of four.
In contrast, the luminous effectiveness (brightness or luminous flux) only decreases to about 70%. It’s been cut by around a third as a result.








