Experiments show that ocean plastic pollution not only has direct negative effects on marine life but also leads to ocean acidification. The pH of seawater is shown to be significantly lowered due to the release of organic acids and carbon dioxide from plastics when exposed to light. Ocean acidification is already on the rise due to climate change, which is much worse in marine regions that are extensively contaminated with microplastics.
More than 13 million tons of plastic enter the seas every year. They are breaking down into microplastics and traveling with ocean currents to the most inaccessible places on Earth. More than 1.8 trillion pieces of plastic are thought to be floating in the Great Pacific Garbage Patch alone, and this figure is expected to grow. Threatening marine life and wildlife, plastic pollution also releases toxins and creates greenhouse gases like methane and carbon dioxide as it decomposes.
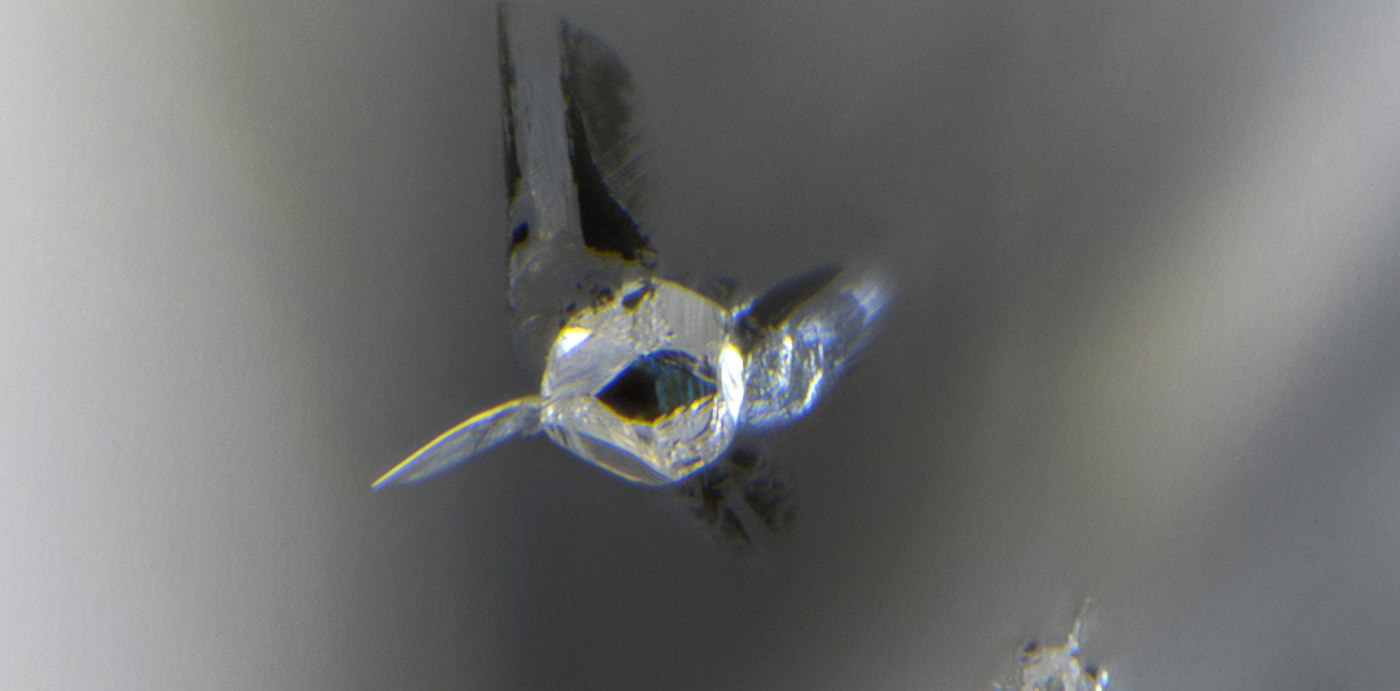
An experiment on the breakdown of plastic in the ocean
Now, researchers led by Cristina Romera-Castillo of Barcelona’s Institute of Marine Sciences have discovered another negative effect of plastic waste. They were curious as to how the acidity of the water is affected by the photochemical breakdown of plastic debris at sea. It was previously recognized that this leaching process may change the biogeochemistry of the ocean because plastic releases dissolved organic materials. This includes chemicals like carbon dioxide (CO2) and organic acids, which may both have a net negative effect on the pH of their surroundings.
In test containers with pure seawater, the researchers introduced several kinds of plastic that had been crushed to a size of only a few millimeters to see whether this also occurred during the decomposition of plastic in the ocean. Polyethylene film fragments—the most frequent kind of plastic trash—alongside expanded polystyrene and bioplastic polylactic acid with a variety of ages and compositions were collected for the experiment from the beach.
There were around 78 plastic pieces per liter, on average. That’s the population density expected to be found in a badly polluted coastal area. The pH of a test batch of ale was determined before and after being exposed to artificial sunshine for six days.
Acidification has a stronger impact than climate change
The pH of the ocean water dropped significantly due to the breakdown of the old plastic and expanded polystyrene. The pH of the water decreased from 0.33 to 0.54 after being exposed to sunshine for a few days. Fresh polyethylene film and bioplastic, on the other hand, had a chemical influence on the water that was less noticeable, increasing the water’s acidity by just 0.02 to 0.03 units in six days.
Climate change increases atmospheric and oceanic CO2 concentrations, which result in more acidification and reduce ocean pH by an average of 0.02 pH units every decade. The research found that plastic waste might have an acidifying impact on the ocean that is even more severe than the ocean acidification predicted for certain climatic scenarios by the end of this century. Ocean acidification brought on by human-produced CO2 will be made worse by the decomposition of this plastic, which will cause a drop in pH.
Sunlight’s ultraviolet (UV) rays, in particular, have been shown to accelerate the chemical breakdown of plastics, as was validated by the researchers’ closer inspection. These decompose, releasing organic acids and CO2, which might further acidify the water.
A significant issue for coastal waters
A major cause for worry, according to the scientists, is that these acids are released in disproportionately high concentrations by plastic that has been floating in the water for longer. Ocean plastic is often comprised of a variety of polymers that have all aged due to exposure to chemical weathering over time. In particular, this plastic debris burden has the potential to drastically alter the chemistry of saltwater in extensively contaminated coastal locations.
Based on the results, it seems that plastic breakdown significantly decreases pH levels. Some aquatic life may temporarily have their tolerance levels exceeded as a consequence of the acidification. (Science of The Total Environment, 2022; doi: 10.1016/j.scitotenv.2022.158683)