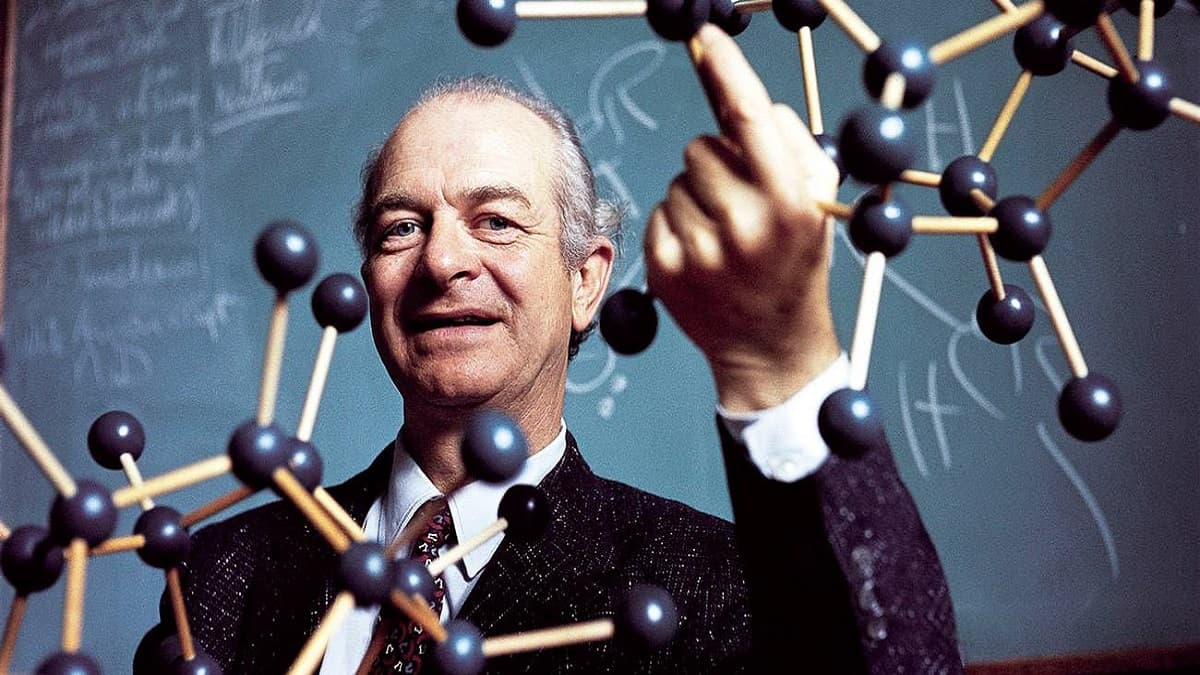Chemistry dominated Linus Carl Pauling’s (or Linus Pauling) entire spiritual and social life, beginning from his youth when he first witnessed a chemical reaction at a friend’s house in Oregon until his final months on his farm in Big Sur Beach, California. When he proposed to his future spouse, he was honest enough to say that marriage would come after his job. Linus Carl Pauling’s passion for science paid off; he made discoveries about the nature of chemical bonds and the basic structure of important biological molecules such as proteins.
Who was Linus Carl Pauling?
In 1954, Linus Carl Pauling was awarded the Nobel Prize in Chemistry for these discoveries. His scientific expertise also supported his humanitarian efforts. The evidence he provided that fallout from above-ground nuclear tests caused a large number of birth defects and cancers was the main factor in his 1962 Nobel Peace Prize. The award was given on October 10, 1963, on the day the Partial Nuclear Test Ban Treaty entered into force. Linus Pauling became the first person in history to receive two Nobel Prizes on his own.
Linus Carl Pauling was the eldest and only son of three children of pharmacist Herman W. Pauling and Lucy Isabelle Pauling, also the daughter of a pharmacist. Linus spent the first years of his life in Condon, in the monotonous western town of Oregon’s hinterland, where his father’s pharmacy was located. His childhood memories included cowboys, of whom one taught him how to sharpen a pencil with a knife, and Native Americans who showed him how to find and dig edible roots. These seemingly negligible lessons taught him in two ways: there was the right technique to do a job, and experienced people were valuable sources of information.
In the primitive elementary school in Condon, his favorite subjects were arithmetic and spelling because they were only interested in right or wrong answers. Economic difficulties and the fire in the shop caused Herman to move his family to Portland in 1909. Not long after starting a new pharmacy, Pauling’s father died suddenly at the age of 33 of a perforated stomach ulcer.
His mother, who lacked any profitable skills, borrowed heavily to buy a large house in hopes that it would enable boarders and room renters to support her and her children, but she was often in short supply of money and had health problems. Linus had to work at jobs where he delivered milk and newspapers. When he became interested in chemistry, he set up a laboratory in the basement and started doing simple experiments.
Linus Carl Pauling and his extraordinary educational background

He also took all the science and mathematics courses he could at Washington High School but left without a diploma because he had not taken the mandatory American history courses (instead of those courses, he took mathematics). He later had a well-paid job in a workshop that produced cargo elevators. Her mother wanted her to give up her university plans and continue to support the family. Luckily, Linus’s friend’s father stepped in and made it possible for Belle Pauling to let her son go to OAC, which was now Oregon State University.
Linus Pauling continued his studies in chemical engineering (the only major available for prospective chemists at the OAC) with extraordinary success, while at the same time working in a variety of jobs to support himself, his mother, and his sister. He even had to drop out of school for a year due to his mother’s financial problems. He worked as a road construction supervisor at that time, then became a quantitative analysis assistant at OAC. Around this time, he began reading articles by Gilbert Newton Lewis and Irving Langmuir on chemical bonds. While teaching chemistry to female home economics students in his senior year of school, he met his future wife, Ava Helen Miller.
After graduating from the OAC in 1922, Pauling began his graduate education at the California Institute of Technology (commonly known as Caltech or Pauling’s preferred acronym, CIT). Along with the weighty courses he took, Linus Pauling began research under the supervision of X-ray crystallographer Roscoe Gilkey Dickinson, who directed him to the structural studies of the mineral molybdenite. They wrote an article about it because sulfur atoms in the mineral molybdenite were found to be arranged in a triangular prismatic shape around molybdenum atoms.
Linus Pauling married Ava Helen after her first year at CIT; his husband became the main supporter of scientific research and peace efforts in the ensuing years and held this role throughout their fifty years of marriage. Pauling successfully defended his dissertation, which was based on articles about crystal structure, and got his Ph.D. in 1925.
Determining the nature of chemical bonds
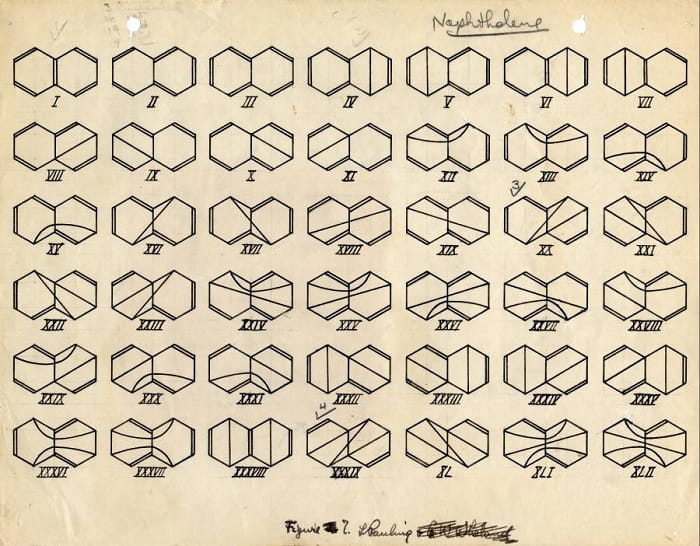
In 1926, he received the Guggenheim Fellowship and went to Europe with his wife. There he explored the possible effects of newly discovered quantum mechanics on his work on the nature of chemical bonds—the gravitational forces that hold atoms together in a unified form. Although he also spent time at Niels Bohr’s Institute in Copenhagen and with Schrödinger at the University of Zurich, he was most influenced by the Arnold Sommerfeld Center for Theoretical Physics in Munich. He started making predictions about the properties of ionic crystals using wave mechanics, Sommerfeld’s favorite type of quantum mechanics.
Linus Pauling returned to CIT in 1927 and began his long and successful career in X-ray studies of crystal structures such as silicate minerals. His work on crystal structures helped make this branch one of the best understood in science. Using what he knew about bond angles and distances, he came up with rules called “coordination theory” that would make it easier for crystallographers to put the right atoms in the right places in different crystals.
During a meeting with Herman Mark in Germany in 1930, electron diffraction caught his attention. Using this technique, he and his colleagues solved the structures of many molecules in gas and liquid.
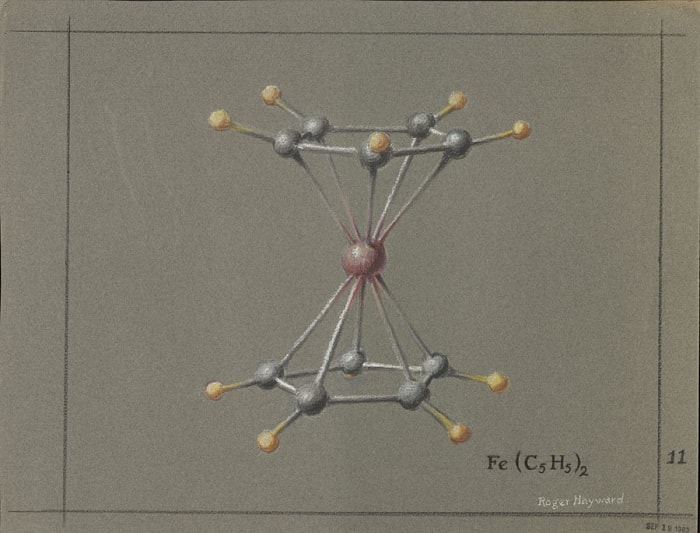

In the 1930s, he used the energy of the displacement (or oscillation) of two electrons in the hybridization analysis (involving the mixing of atomic orbitals—the location of a particular electron in an atom). It was a revolutionary idea featured in his best-known articles on the nature of chemical bonds. Linus Pauling’s knowledge of quantum mechanics was a big part of how he came up with the valence bond theory. In this theory, he said that some molecules, like benzene, could be thought of as intermediates made up of hybrids of two or more structures in which atomic orbits overlap.
The Nature of Chemical Bonds and the Structure of Molecules and Crystals, a book he wrote in 1939 based on the George Fisher Baker Lectures at Cornell University, is a summary of his own experimental and theoretical work as well as that of other structural chemists.
In the mid-1930s, Linus Pauling’s interest began to shift to biological molecules; he and his colleagues conducted magnetic studies on hemoglobin to prove that the magnet attracts hemoglobin in veins but repels it in arteries. Hemoglobin was the protein molecule. That work inevitably led him to be more generally interested in proteins, such as the roles of proteins in the antibody-antigen response. He worked on the denaturation of proteins and the human immune system’s antibodies, which fight against antigens that attack in the form of bacteria or viruses.
The discovery of the first molecular disease

During World War II, Linus Pauling started to focus on more practical problems, such as providing plasma supplies for wounded soldiers by producing an artificial mixture that could be used in place of blood plasma. He invented the oxygen detector based on the magnetic properties of oxygen particles. This invention was widely used in submarines and aircraft. He had also worked on explosives, rocket fuel, and ink for confidential correspondence. He refused J. Robert Oppenheimer’s offer to lead the chemistry-related arm of the atomic bomb project as he was battling a serious disease called glomerulonephritis. Towards the end of the war, Linus Pauling learned about the inherited disease sickle-cell anemia, where the red blood cells in venous blood take the shape of a sickle. He thought that this sickle shape was caused by a genetic mutation in the globin part of the cell’s hemoglobin. After three years of study, Pauling and colleagues were able to prove that such a molecular defect in hemoglobin was indeed the cause of the disease. Thus, Linus Pauling discovered the first molecular disease.
In the post-war years, Linus Pauling continued to work on proteins. In the early 1950s, he published a cylindrical helix amino acid arrangement of amino acid groups (later called alpha helices) linked by hydrogen bonds. These and other protein structures he published were extremely effective. In addition to that work, Linus Pauling also participated in educating the public about the possible consequences of nuclear weapons. He devoted more and more time to campaigns to stop nuclear weapons testing in the atmosphere.
In January 1958, Linus Pauling and his wife submitted to the United Nations an application signed by more than 9,000 scientists to stop the trials.
Although United States officials wanted to undermine his efforts by confiscating his passport, they had to return it when he won the Nobel Prize in Chemistry in 1954. Throughout the rest of the 1950s and 1960s, Pauling and his wife tried to spread the word about this case around the world. For his efforts, Pauling won the Nobel Peace Prize in 1963 (his wife did not share the prize as the Nobel bureaucracy was mostly male and had not nominated her).
Second Nobel and Vitamin C

Linus Carl Pauling left the Institute in 1963 as a result of the CIT representatives’ negative response to his peace studies and the Nobel Peace Prize. The laboratory space Pauling used for molecular medicine studies was taken away to punish him. In the mid-1960s, he was working at the Center for the Study of Democratic Institutions. His humanitarian work was supported here; he developed a theory of atomic nuclei (the theory was eventually rejected by many nuclear physicists). He needed a laboratory for his experimental research, and in 1967 he became a professor of chemistry at the University of California, San Diego. Here he dealt with the neglected potential of vitamin C to cure diseases such as the common cold. He accepted the professorship at Stanford in 1969. In 1970, he published a book called Vitamin C and the Common Cold, which had the largest readership and initiated a debate over megavitamin therapy that lasted until the end of his life.
His view on the effects of using large amounts of vitamin C in the treatment of infections, cancer, and other diseases had been largely rejected by medical institutions. Pauling co-founded the Institute for Orthomolecular Medicine in 1973. It was later renamed the Linus Pauling Institute of Science and Medicine, and its main goal was to find evidence in the lab and in real life to backup Pauling’s ideas.
Linus Carl Pauling’s death and view on life
Linus Pauling’s institute grappled with personal and legal issues, and he faced further challenges after his wife’s death in 1981 and when he was diagnosed with prostate cancer in 1991. Despite these problems, Linus Pauling keeps working. He developed arguments, in particular, against crystallographers who violated the traditional rules he defended by considering quintet symmetric semi-crystals intrinsic.
What he did in the last 20 years was no different than the years when he hung up tables of chemical substances and their properties on the laboratory desk in the basement of his mother’s boarding house: that is, to investigate the connections between the structures and functions of molecules not only with chemistry but with physics, biology, and medicine.
As an atheist and reductionist, he believed in the power of science to answer any questions people might ask. For him, the universe consisted of only matter and energy. The structure of molecules had the potential to explain all physical, chemical, biological, and even psychological phenomena. He talked about the death of his wife and how it made him feel. At the same time, his cancer spread from his prostate to his intestines and then to his liver, killing him in 1994.
He left behind a unique body of chemical knowledge that he said would lead to wealth, variety, and new discoveries.
Bibliography:
- Marinacci, Barbara; Krishnamurthy, Ramesh (1998). Linus Pauling on Peace. Rising Star Press. ISBN 978-0-933670-03-7.
- Serafini, Anthony (1989). Linus Pauling: A Man and His Science. Paragon House. ISBN 978-1-55778-440-7.
- Linus Pauling at the Mathematics Genealogy Project
- A Guggenheim Fellow in Europe during the Golden Years of Physics (1926–1927)”. Oregon State University. Archived.